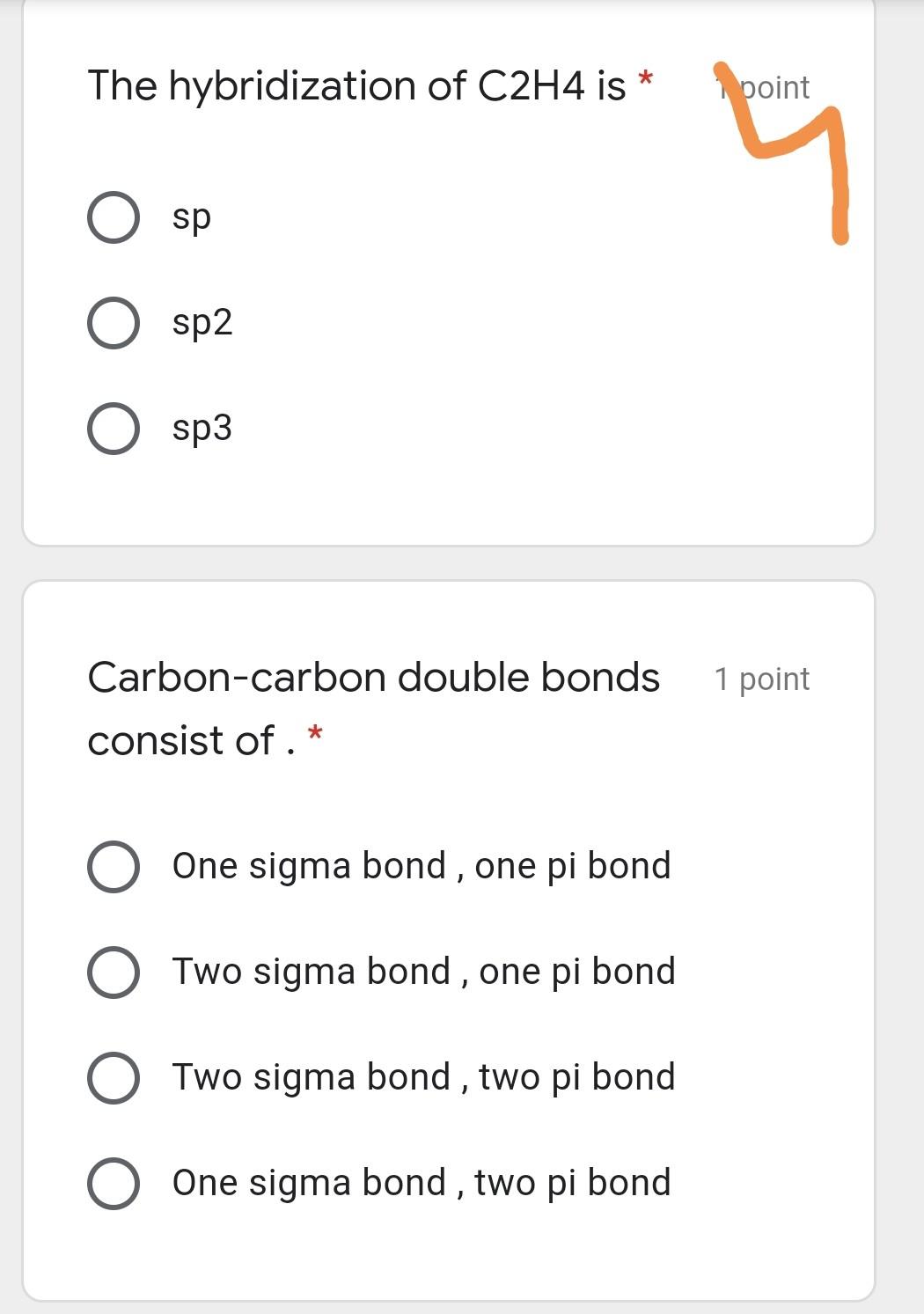
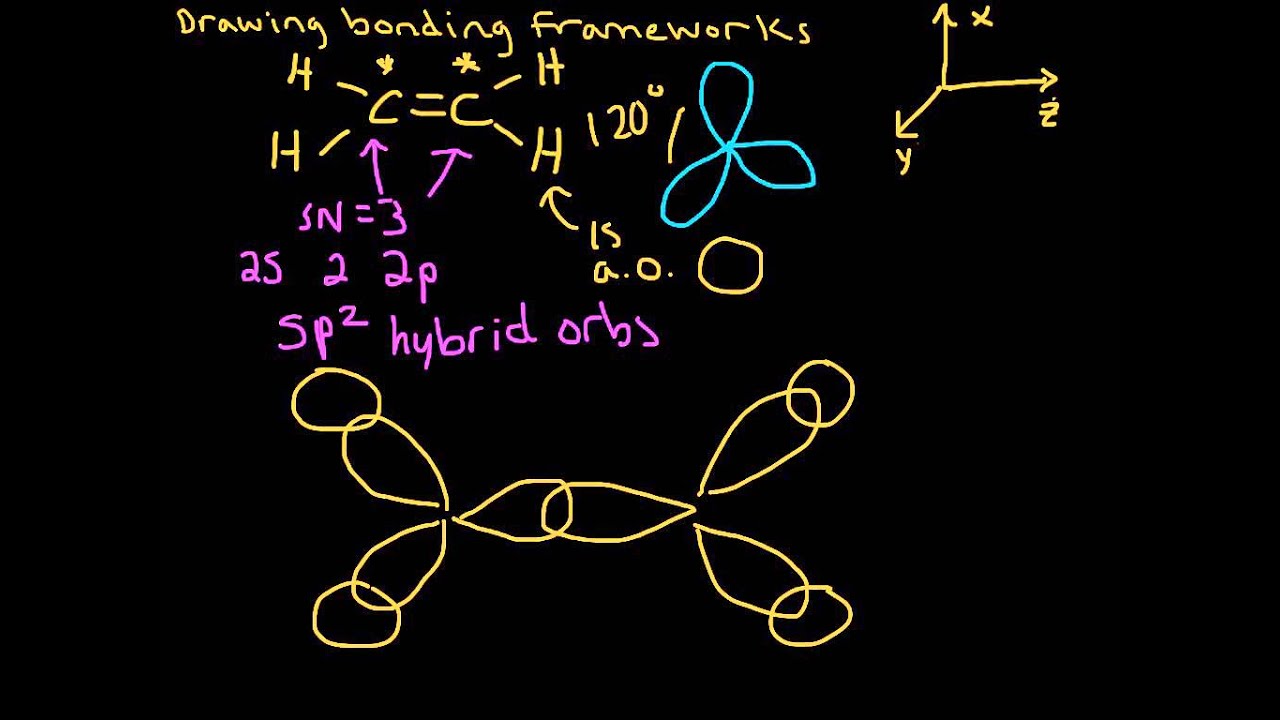
Hybridized orbital energy level diagrams for a) H2; b) C₂H₆; c) C2H4;... | Download Scientific Diagram

Please explain sp2 hybridisation in C2H4 and sp hybridisation in C2H2, with ground state and excited state diagrams for carbon - Chemistry - Chemical bonding and molecular structure - 8758411 | Meritnation.com

molecular orbital picture of ethylene, formation of sp2 hybridisation. 28282149 Vector Art at Vecteezy
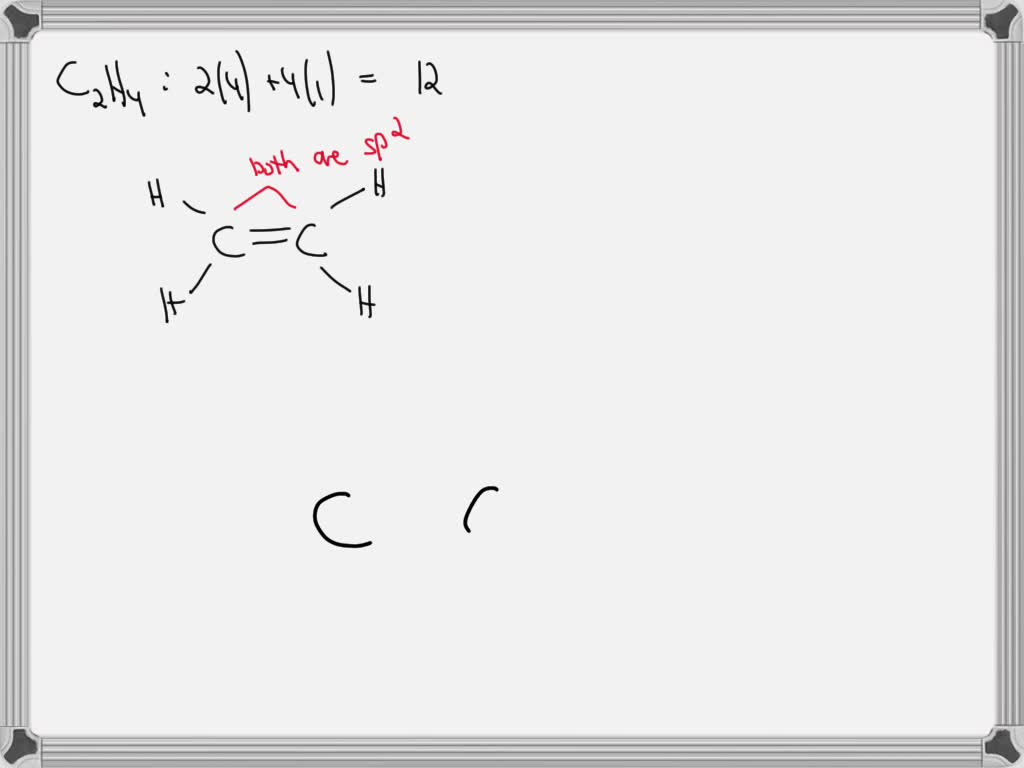
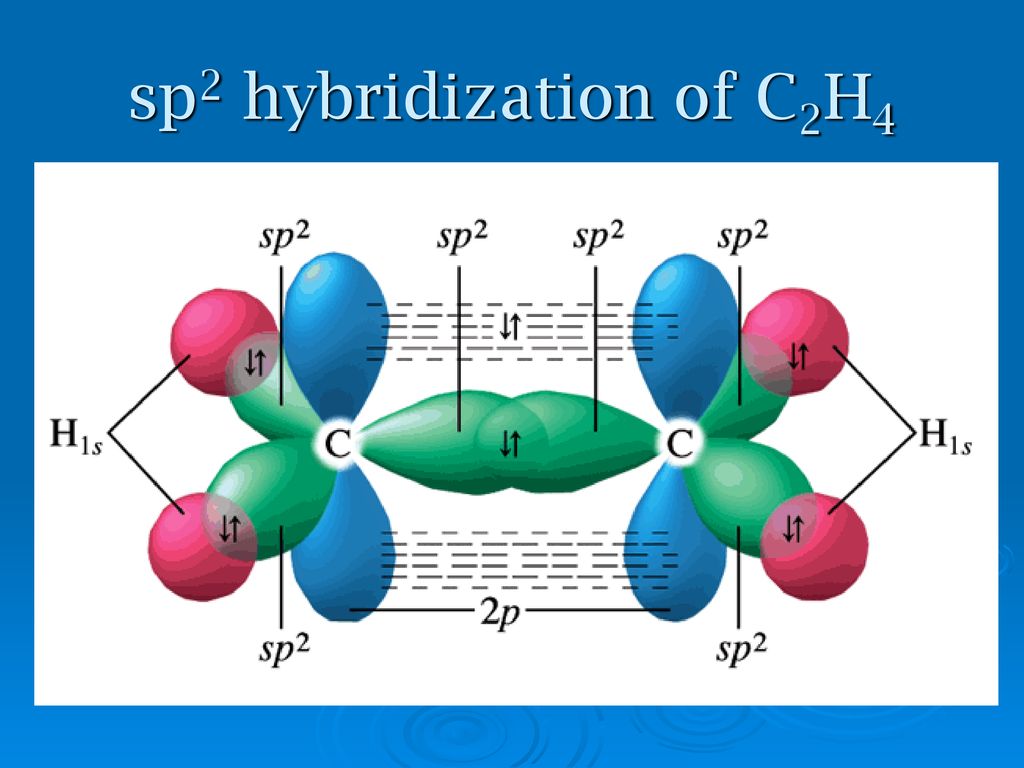
SOLVED: Apply Valence Bond (VB) theory to the ethylene (C2H4) molecule, and: i) Identify the type of hybridization of carbon atoms in C2H4 (justify your answer by drawing the promotion/hybridization scheme for
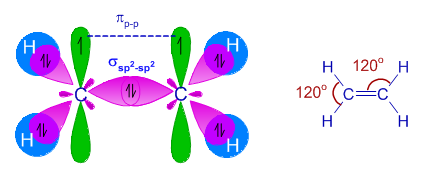
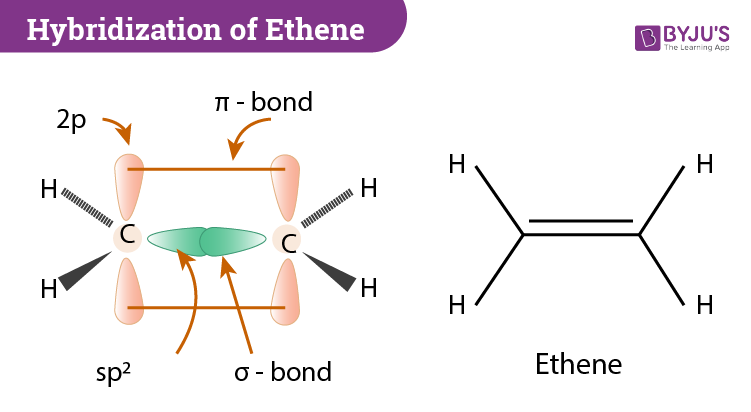
Explain sp^2 hybridization in ethane molecule. C2H4 – ethene. - Sarthaks eConnect | Largest Online Education Community
Why didn't the 3rd P orbital of carbon in C2H4 take part in hybridization as it took part in Hybridization in CH4? - Quora

Hybridization|sp3,sp2,sp hybridization |Hybridization of carbon,nitrogen| c2h4,c2h2,nh3 hybridization | chemistry, nitrogen, carbon | Hybridization is the mixing up of atomic orbitals to form new orbitals. The orbital hybridization may be of different ...