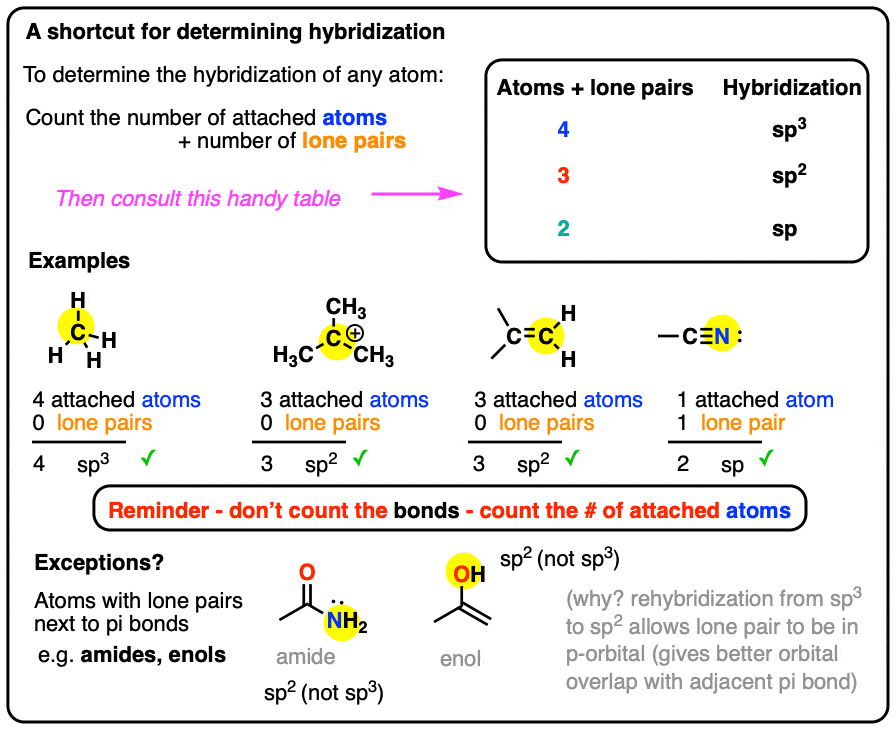
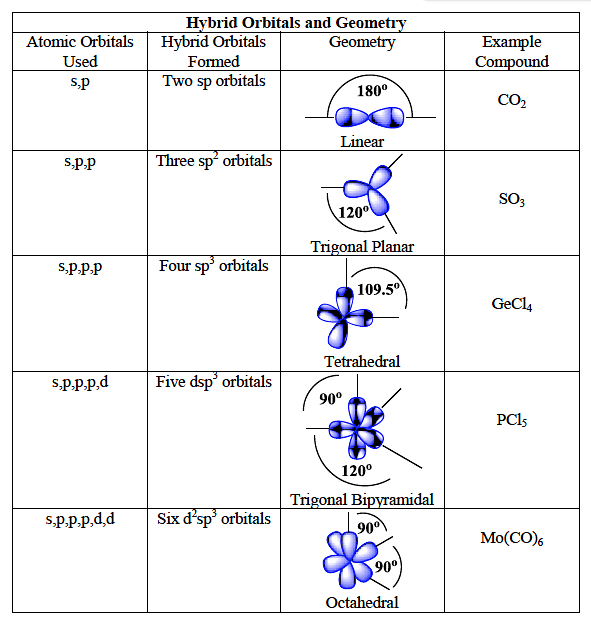
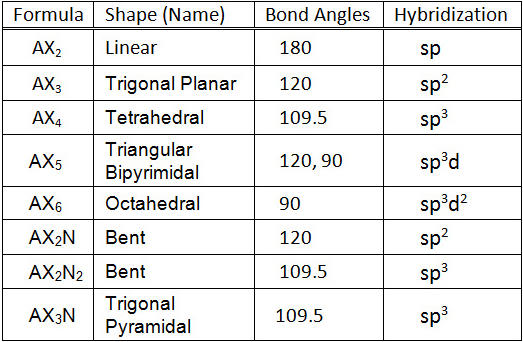
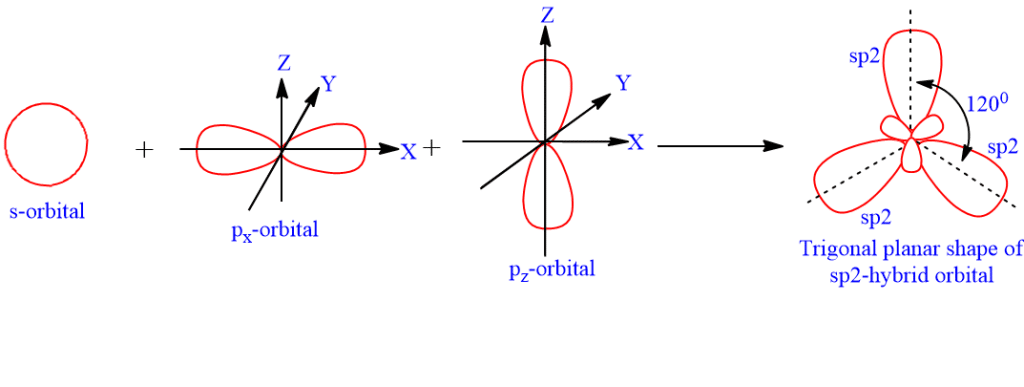
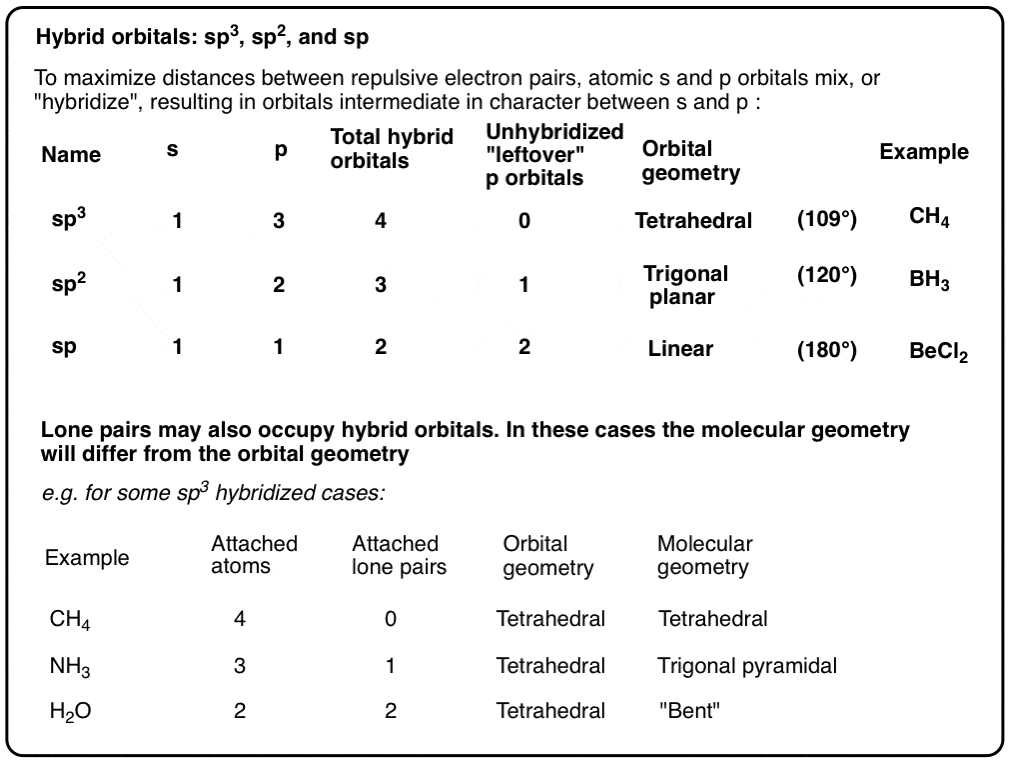
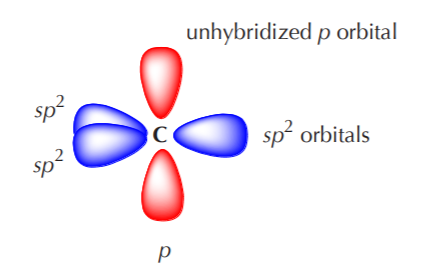
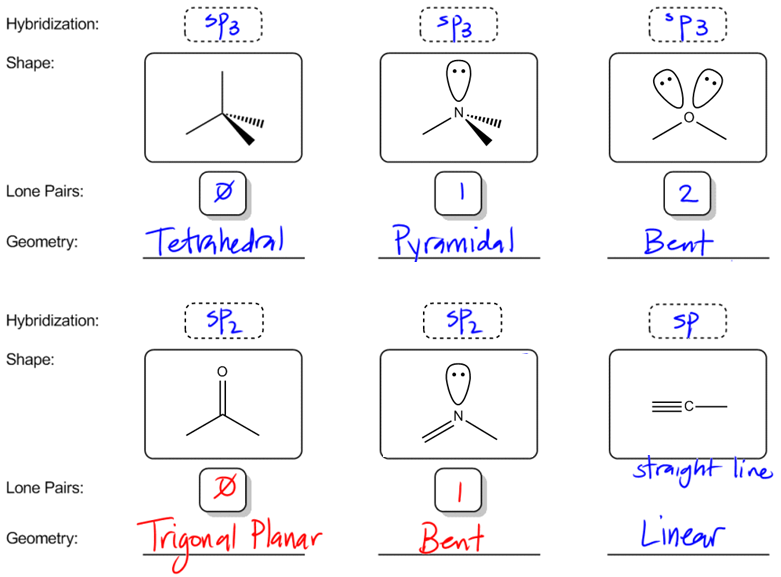
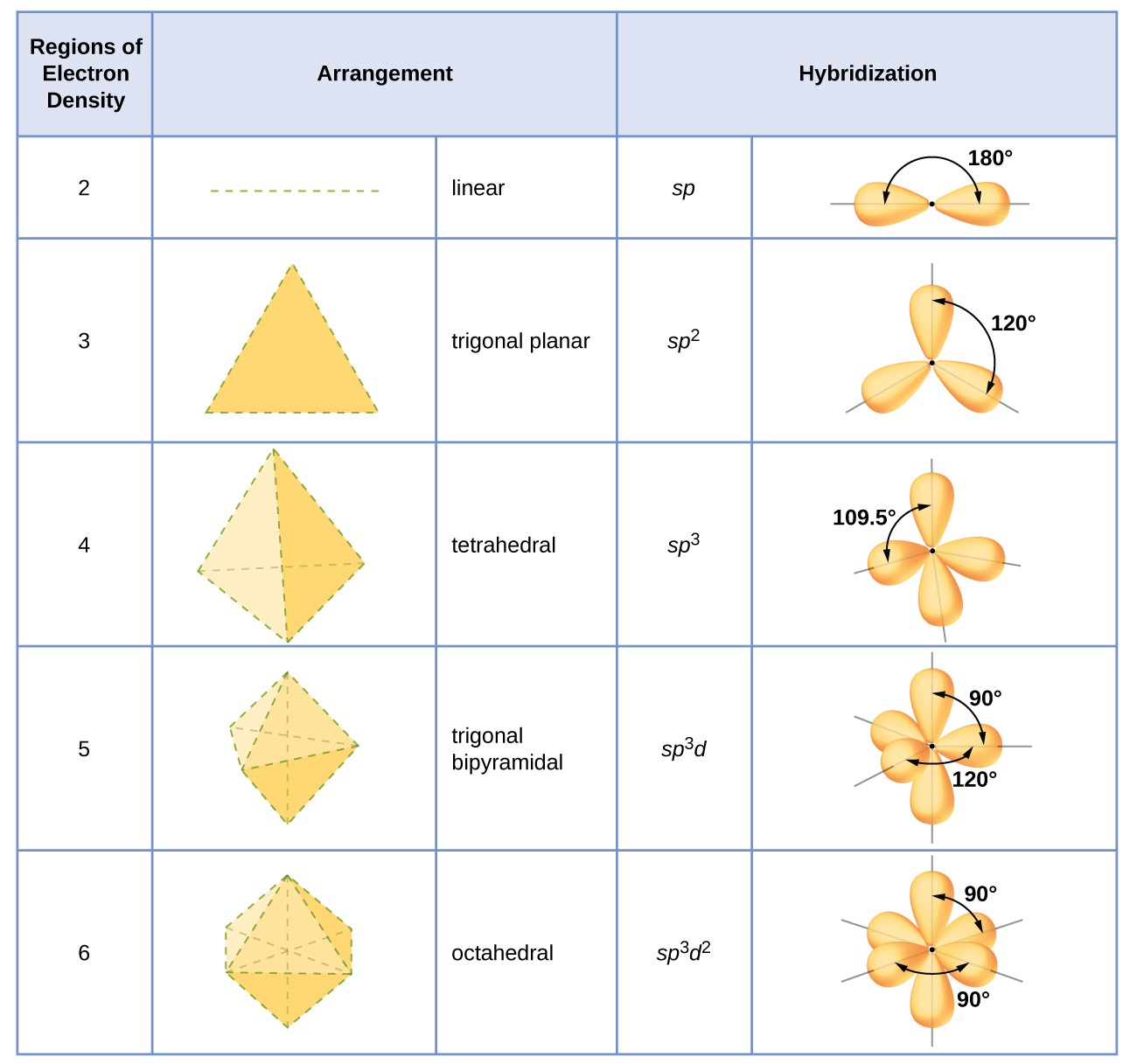
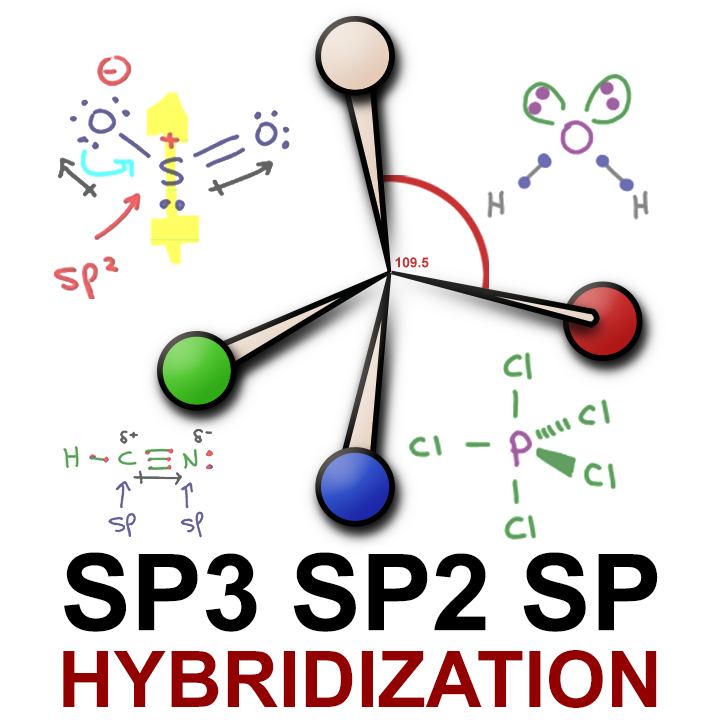
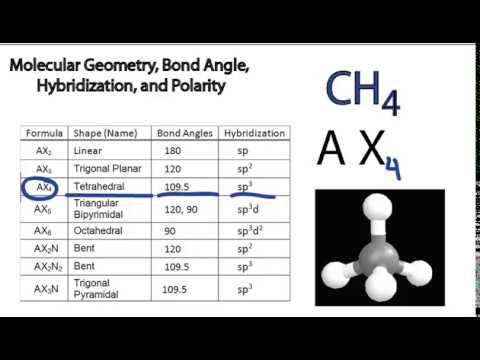
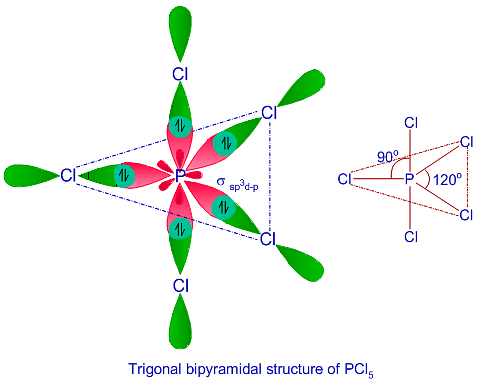
What are the shapes and bond angles of sp, sp2, sp3, sp3d, sp3d2 hybridised orbitals respectively? - Quora

SOLVED: What is the hybridization and bond angle of each of the carbons in this compound? The carbons in the answer should be reported from left to right: As before, do not
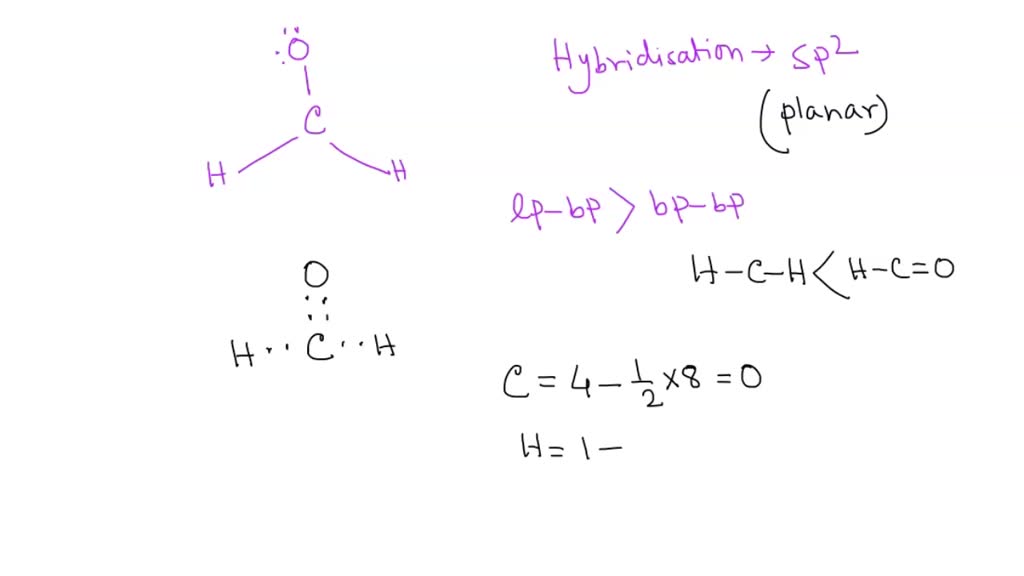
SOLVED: What is true about the CH2O molecule? Carbon utilizes sp2 hybridization. The H-C-H bond angle is expected to be slightly less than the H-C-O bond angle. The molecule contains 3 sigma
What are the shapes and bond angles of sp, sp2, sp3, sp3d, sp3d2 hybridised orbitals respectively? - Quora
What are the shapes and bond angles of sp, sp2, sp3, sp3d, sp3d2 hybridised orbitals respectively? - Quora
What are the shapes and bond angles of sp, sp2, sp3, sp3d, sp3d2 hybridised orbitals respectively? - Quora