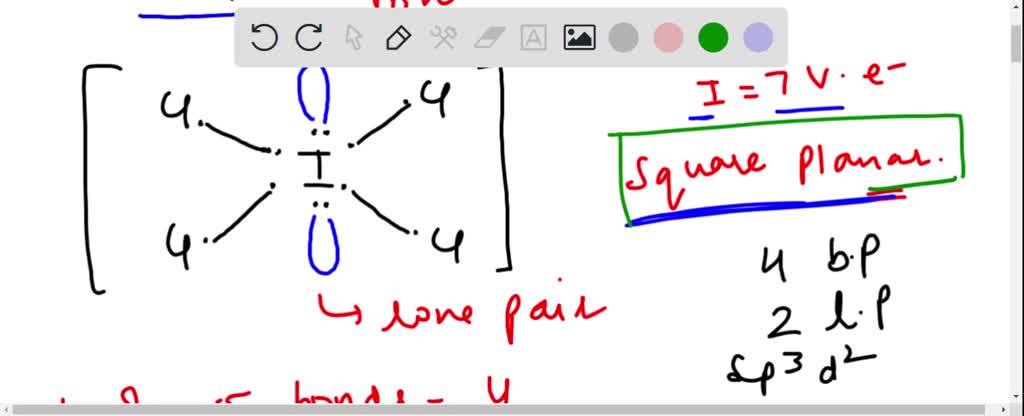
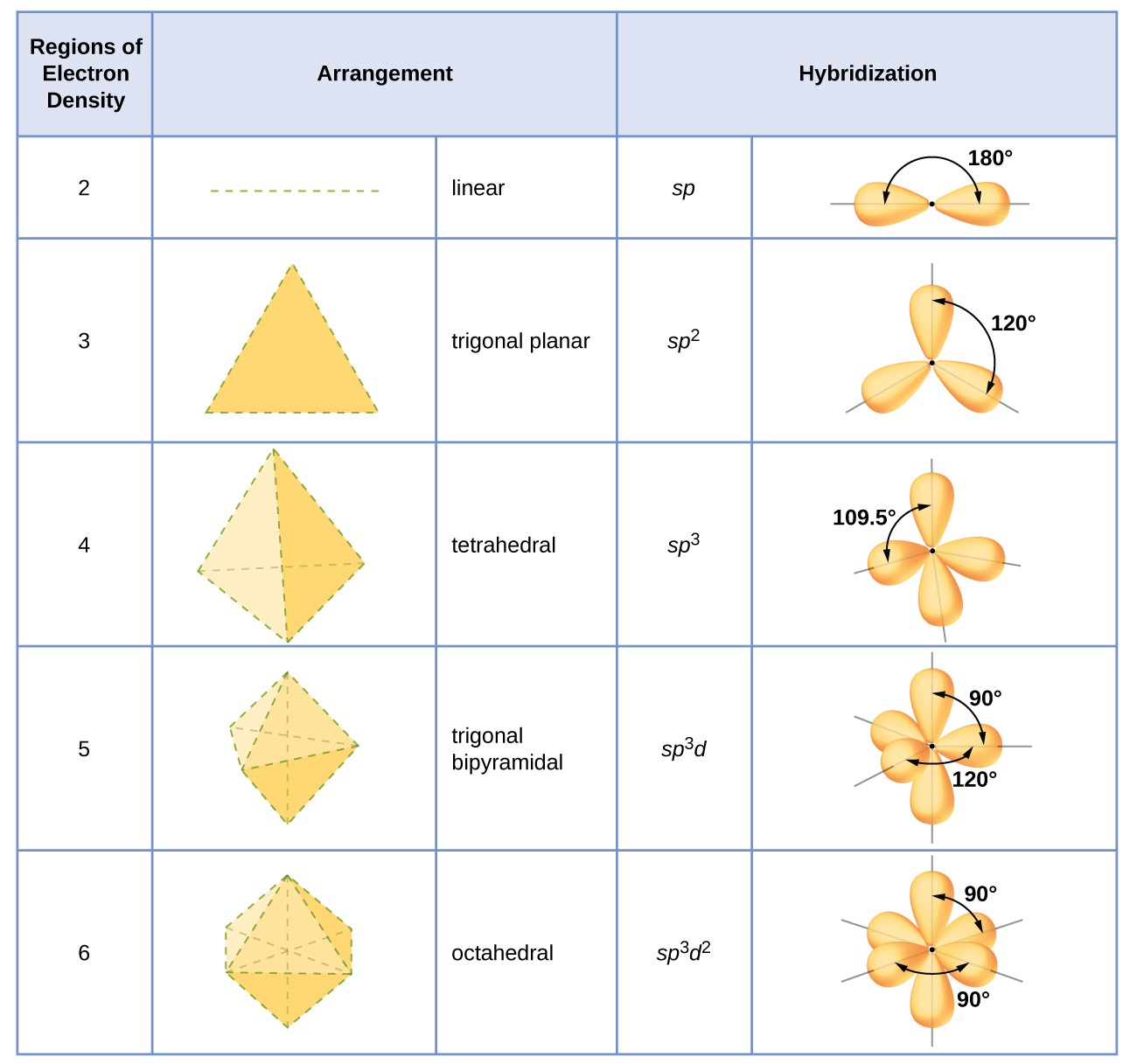
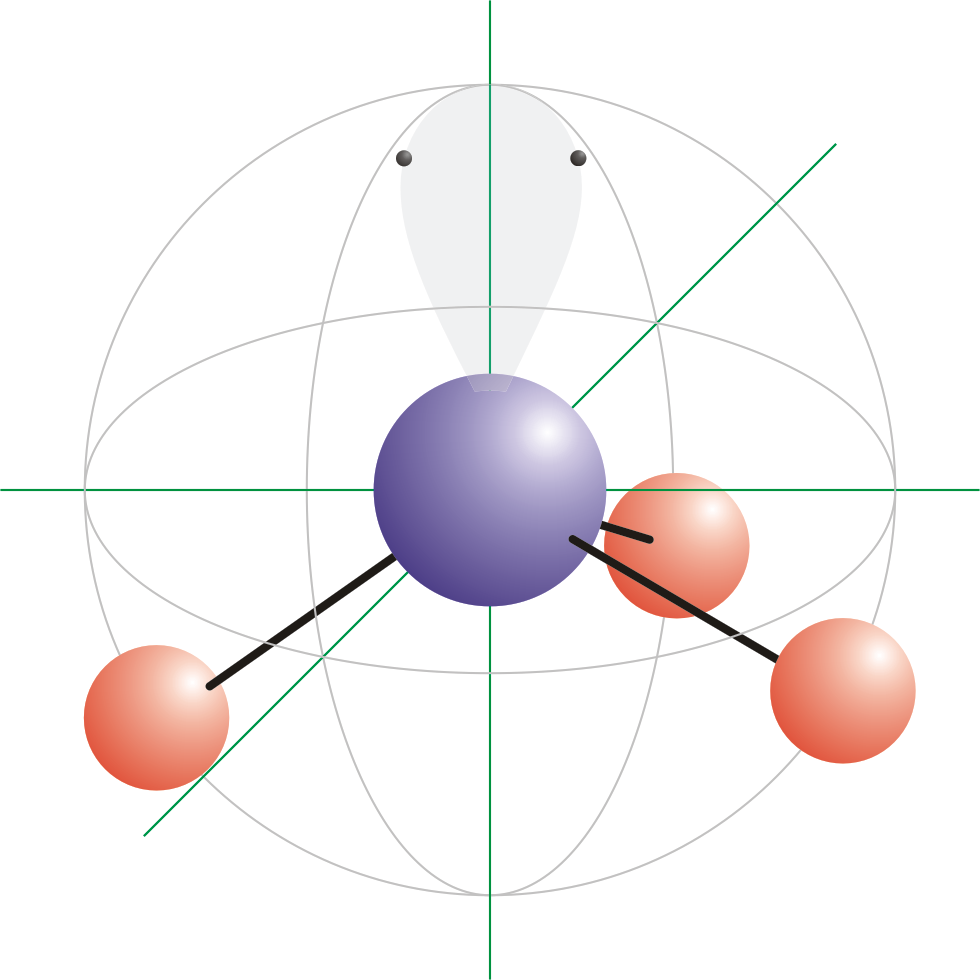
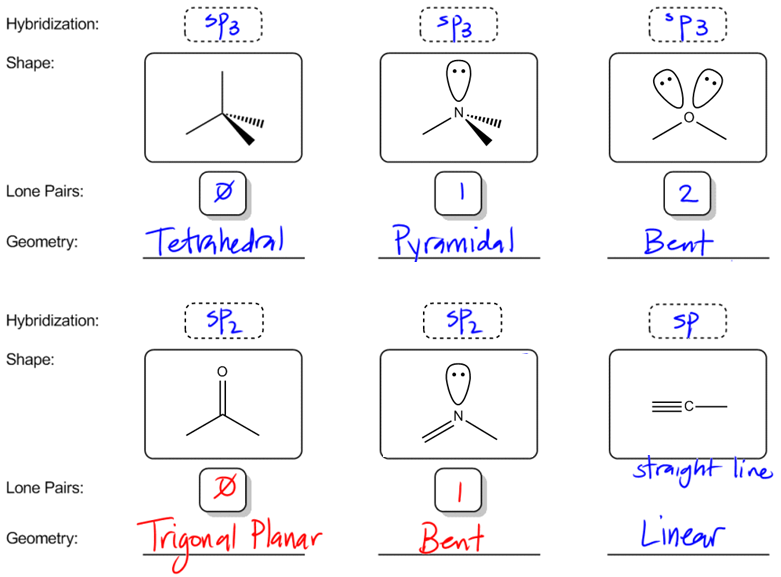
A complex involvong dsp^{2} hybridization has :a tetrahedral geometryan octahedral geometrya square planar geometrytrigonal planar geometry

Match the compounds given in column I with the hybridization and shape given in column II and mark the correct option.

Sir if hybridization is dsp2 then geometry is square planar and if hybridization is sp3d2 then shape is square planar - Chemistry - Coordination Compounds - 13366877 | Meritnation.com
![inorganic chemistry - Why is [PdCl4]2- square planar whereas [NiCl4]2- is tetrahedral? - Chemistry Stack Exchange inorganic chemistry - Why is [PdCl4]2- square planar whereas [NiCl4]2- is tetrahedral? - Chemistry Stack Exchange](https://i.stack.imgur.com/xHv3g.png)
inorganic chemistry - Why is [PdCl4]2- square planar whereas [NiCl4]2- is tetrahedral? - Chemistry Stack Exchange

For each atom listed, identify the geometry as one of the following: tetrahedral, square planar, trigonal planar, trigonal pyramidal, or linear. | Homework.Study.com

Valence bond theory of Coordination Compounds- Features, Hybridisation, Geometry, Examples, Limitation and FAQs of Valence bond theory.

Odes it make any difference if we write hybridisation as sp3d2 or d2sp3 Shape of molecules/ ions Square planar - Chemistry - Chemical Bonding and Molecular Structure - 13299455 | Meritnation.com

A square planar complex is formed by hybridization P of which atomic orbitals W (1) \( s, p_{x}... - YouTube
![According to the valence bond theory, the hybridization of central metal atom is dsp2for which of the following compounds?a)Na2[NiCl4]b)NiCl2.6H2Oc)K2[Ni(CN)4]d)[Ni(CO)4]Correct answer is option 'C'. Can you explain this answer? - EduRev JEE Question According to the valence bond theory, the hybridization of central metal atom is dsp2for which of the following compounds?a)Na2[NiCl4]b)NiCl2.6H2Oc)K2[Ni(CN)4]d)[Ni(CO)4]Correct answer is option 'C'. Can you explain this answer? - EduRev JEE Question](https://edurev.gumlet.io/ApplicationImages/Temp/003bd7dd-46f9-4901-8626-8f092d259fd3_lg.jpg)
According to the valence bond theory, the hybridization of central metal atom is dsp2for which of the following compounds?a)Na2[NiCl4]b)NiCl2.6H2Oc)K2[Ni(CN)4]d)[Ni(CO)4]Correct answer is option 'C'. Can you explain this answer? - EduRev JEE Question